A major development in medical research is raising new hope for patients battling one of the most aggressive forms of breast cancer. A late-stage clinical trial evaluating an innovative targeted therapy has reported promising results, potentially paving the way for a new treatment option for patients with metastatic triple-negative breast cancer. Scientists say the findings represent an important step forward in the fight against a disease that has historically been difficult to treat.
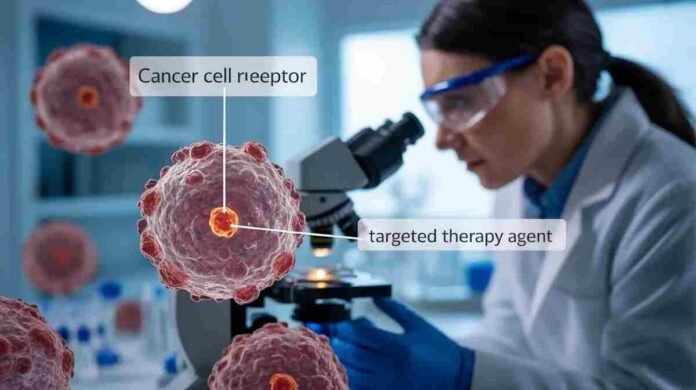
The experimental therapy, known as izalontamab brengitecan, belongs to a new class of medicines called antibody-drug conjugates. These treatments combine targeted antibodies with powerful chemotherapy drugs, allowing doctors to deliver cancer-killing compounds directly to tumor cells while limiting damage to healthy tissue. Researchers believe this targeted approach could significantly improve outcomes for patients whose cancers do not respond to conventional treatments.
The drug recently achieved positive results in a Phase 3 clinical trial involving patients with advanced or metastatic triple-negative breast cancer. In the study, the therapy demonstrated significant improvements in both progression-free survival and overall survival compared with standard chemotherapy options chosen by physicians. These two measurements are among the most important indicators used in oncology research to determine whether a new treatment can truly benefit patients.
Triple-negative breast cancer represents a particularly challenging form of the disease because it lacks three key receptors that many traditional treatments target. Without these receptors, therapies such as hormone treatments and certain targeted drugs are ineffective, leaving chemotherapy as the primary treatment option for many patients. As a result, survival rates for advanced cases have historically been lower compared with other breast cancer types.
The new therapy aims to overcome this challenge by targeting specific proteins found on cancer cells. Once attached to the tumor, the antibody component of the drug delivers a toxic payload that destroys the cancer cell from within. Researchers say this precision-based strategy allows the therapy to attack tumors more effectively while reducing the widespread side effects associated with conventional chemotherapy.
Clinical trials are conducted in several stages before a treatment can receive approval for public use. Phase 3 trials are the final and most extensive stage of testing, typically involving hundreds or thousands of patients across multiple hospitals or research centers. These studies are designed to confirm a treatment’s effectiveness, monitor side effects, and compare it with existing therapies. Successful results often represent the last major hurdle before regulators consider approving a new drug for widespread medical use.
Researchers involved in the study say the results could reshape the treatment landscape for triple-negative breast cancer. Early analyses suggest the drug not only slowed disease progression but also helped extend the overall survival of patients compared with traditional chemotherapy approaches. For patients facing advanced cancer, even modest improvements in survival and quality of life can represent meaningful progress.
The development also reflects a broader shift occurring in oncology research. Over the past decade, scientists have increasingly focused on precision medicine—treatments designed to target specific molecular features of cancer cells. Advances in biotechnology, artificial intelligence, and genomic research have made it possible to identify new therapeutic targets and design drugs that interact with them more effectively.
Pharmaceutical companies and research institutions worldwide are investing heavily in these next-generation therapies. Dozens of new antibody-drug conjugates, immune therapies, and targeted treatments are currently being tested in late-stage clinical trials for various types of cancer. Many experts believe these technologies will redefine how cancer is treated over the next decade.
While further regulatory review and long-term monitoring are still required, the positive Phase 3 results represent a significant milestone. If approved by health authorities, the new therapy could soon provide an additional option for patients who have exhausted other treatments.
For millions of people affected by cancer each year, advances like this highlight the rapid progress being made in medical science. Each successful clinical trial brings researchers one step closer to developing more effective, personalized treatments that could dramatically improve survival and quality of life for patients around the world.